Establishing expiry date for clinical diagnostic reagents
$ 7.00 · 4.6 (309) · In stock

Product shelf life is an essential product performance requirement that, along with other design requirements, is used to determine the safety and efficacy of a clinical diagnostic

Autoverification of test results in the core clinical laboratory
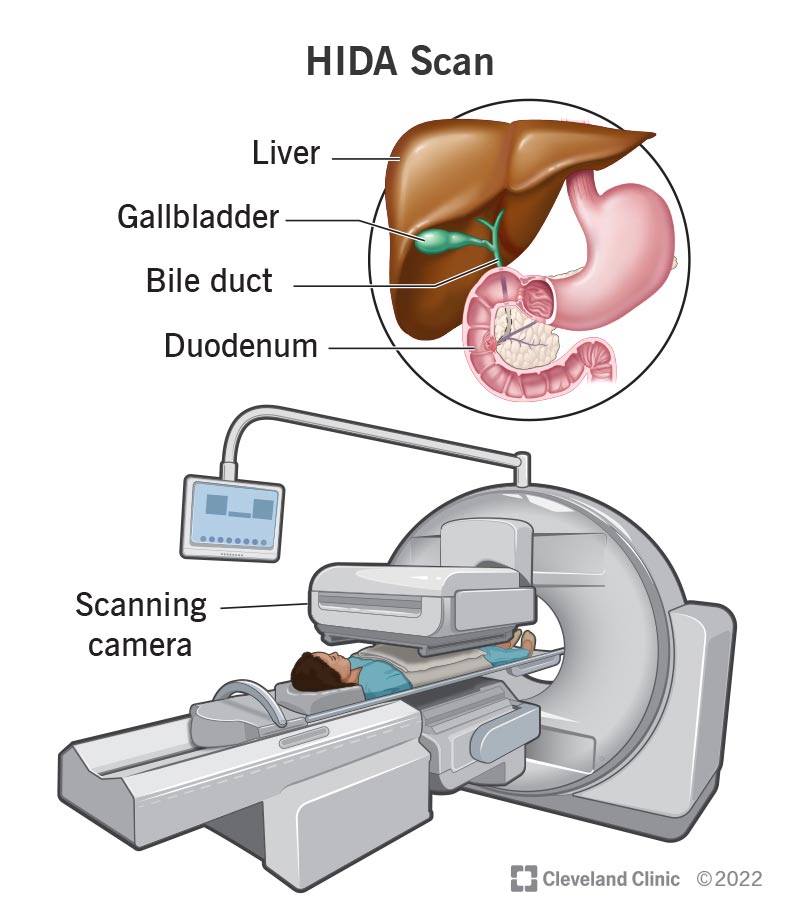
HIDA Scan: What It Is, Purpose, Procedure & Results

UNE EN ISO 23640:2015 In Vitro Diagnostic Medical Devices, 41% OFF

Diagnostic Reagent, Medical Reagents Manufacturer/Supplier
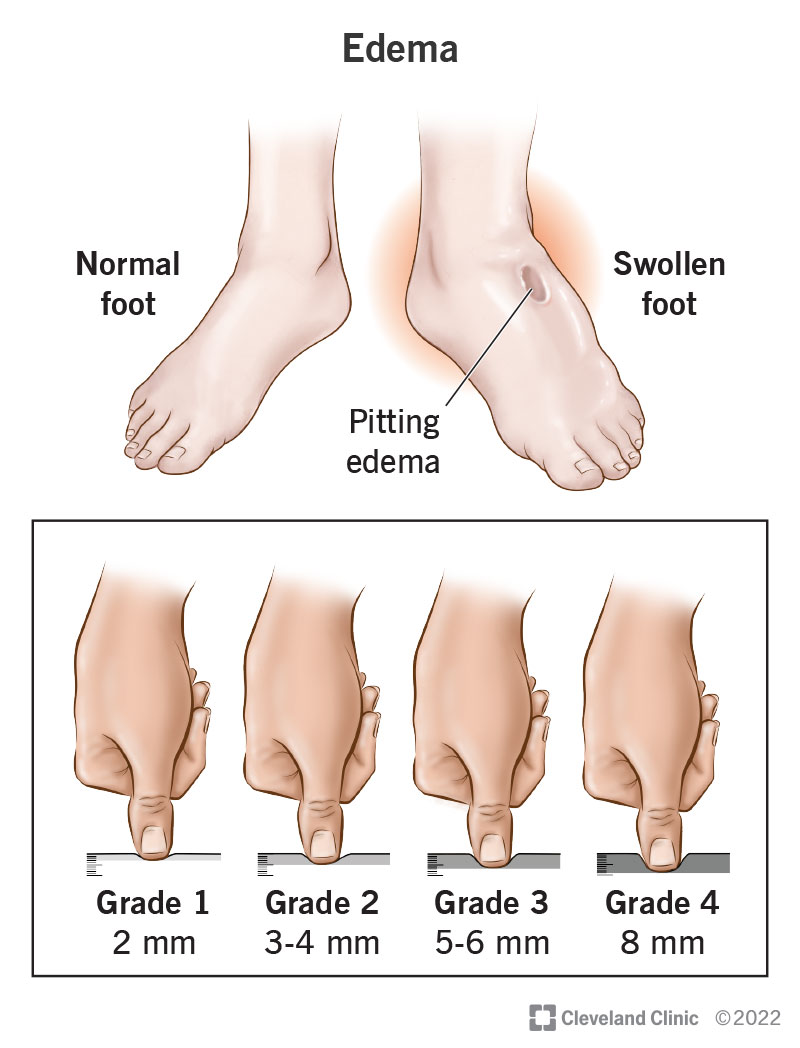
Edema: Causes, Symptoms & Treatment

The Science Behind Food Expiry Dates: Why Consuming Expired Foods

The contributory factors to the scattered picture on AMR

Phases in the development of plant clinics and the Plant Health

An Easy Guide to Chemical Expiry Dates: Top 10 Lab Chemicals
THENA Capital on LinkedIn: #grit #determination #femalefounders
UNE EN ISO 23640:2015 In Vitro Diagnostic Medical Devices, 41% OFF
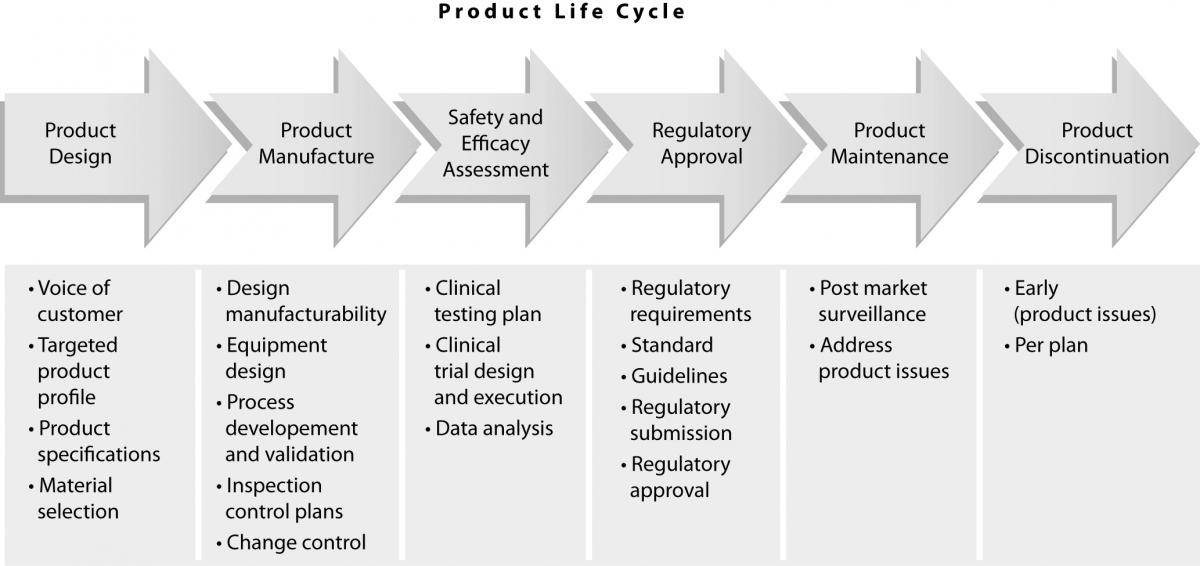
Total quality in laboratory diagnostics: the role of commercial

ASTM F2602-18 - Standard Test Method for Determining the Molar